Biopharma development is a high-stakes game. A single miscalculation in dosage estimation can mess up years of research & cost millions. But the risks go beyond just money—an incorrect dose can lead to severe toxicity, failed trials, and even loss of life. According to Clinical Pharmacology & Therapeutics, 30–40% of drug failures in Phase 1 trials result from poor dose selection.
That’s why Human Equivalent Dose (HED), First-in-Human (FIH) studies, and Maximum Recommended Starting Dose (MRSD) play a critical role in drug safety. They ensure that a drug moves from preclinical models to human trials safely and effectively, reducing the risk of harmful side effects while maximizing therapeutic benefits.
In this blog, we’ll dive into how these calculations work, why they matter, and real-world cases where dosage miscalculations led to devastating consequences.
Understanding Human Equivalent Dose (HED)
HED is a crucial step in drug development. It helps scientists estimate the correct human dosage based on animal studies.
What is HED & Why is it Important?
HED translates drug doses from animals (like rats or monkeys) into equivalent human doses. Since species metabolize drugs differently, direct dose conversion doesn’t work. Instead, researchers use body surface area (BSA) scaling, a method recommended by the U.S. Food and Drug Administration (FDA) for calculating safe starting doses in first-in-human (FIH) trials
How to Calculate HED?
The FDA recommends this formula:
Where K_m values are species-specific constants.
Conversion Factors for Common Test Animals
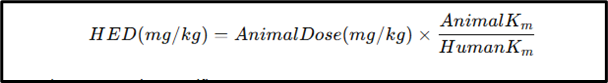
Why Does BSA Scaling Matters?
A 70 kg human isn’t just a scaled-up rat. Metabolic rates differ across species, so BSA-based calculations prevent over- or under-dosing.
Tip: For a deeper understanding, check out this masterclass on determining HED, FIh, and MRSD in biological product development here.
First-In-Human (FIH) Studies: The First Critical Test
FIH studies bridge the gap between preclinical research and human trials.
What is an FIH Study?
First-in-Human (FIH) trials are pivotal in drug development, marking the initial administration of a new compound to humans. These studies primarily aim to assess the drug’s safety, tolerability, and pharmacokinetics (PK). For instance, a recent FIH trial evaluated the safety, tolerability, PK, and pharmacodynamics of zagociguat (CY6463), a central nervous system-penetrant soluble guanylyl cyclase stimulator. The study concluded that single and multiple doses of zagociguat were generally safe and well-tolerated in healthy participants, providing valuable insights into its PK profile.
Case Study
TGN1412 DisasterIn 2006, a monoclonal antibody trial went horribly wrong in the UK. The drug, TGN1412, caused severe immune reactions. 💡 Lesson: Poor dose prediction can be catastrophic. |
Key Phases of FIH Trials
- Phase 0: Microdosing (sub-therapeutic doses to check PK).
- Phase 1: Low-dose trials in small groups (20-100 volunteers).
- Phase 2 & Beyond: Larger trials based on Phase 1 findings.
Challenges in FIH Studies
- Unexpected toxicity: Some drugs behave unpredictably in humans.
- Dosing errors: Too high? Risk toxicity. Too low? Ineffective treatment.
- Ethical concerns: Human safety is non-negotiable.
Maximum Recommended Starting Dose (MRSD): Ensuring Safety
MRSD determines the safest dose to start human trials.
What is MRSD?
The Maximum Recommended Starting Dose (MRSD) is the highest initial dose of a new drug anticipated to be safe for human administration, derived from preclinical studies. Typically, MRSD is calculated from preclinical toxicology studies by determining the No Observed Adverse Effect Level (NOAEL) in animal models, converting this to a Human Equivalent Dose (HED) using body surface area scaling, and then applying a safety factor to account for interspecies differences and uncertainties.
How is MRSD Calculated?
The FDA suggests using the No Observed Adverse Effect Level (NOAEL) in animals:
A standard safety factor of 10 is used to reduce risk.
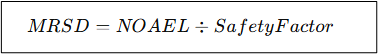
Animal NOAEL (mg/kg) | HED (mg/kg) | MRSD (mg/kg) |
10 (Rat) | 1.62 | 0.162 |
5 (Monkey) | 1.35 | 0.135 |
Why MRSD Matters
Setting the wrong MRSD can lead to:
❌ Toxicity at low doses (risking trial suspension)
❌ Too low doses (wasting time & resources)
The Challenge: Determining the Right Dose for Humans
Transitioning a drug from preclinical trials to human testing is complex. A dose that works in animals might be toxic or ineffective in humans. That’s where HED, FIH, and MRSD calculations help.
- HED converts animal doses to human doses.
- FIH studies test a drug in humans for the first time.
- MRSD ensures a safe starting dose for human trials.
Inaccurate estimations can lead to toxicity, inefficacy, or trial failures. Now, let’s dive deep into how these three factors shape biopharma research.
Final Thoughts: Smarter Drug Development with Expert Guidance
HED, FIH, and MRSD form the foundation of early-stage drug development. These calculations help researchers:
✅ Predict safe and effective doses
✅ Minimize risks in human trials
✅ Improve clinical success rates
As AI and big data reshape dose estimation, the future of biopharma will see faster, safer, and more precise drug development. At MetamorphEDU, we specialize in guiding biotech and pharmaceutical teams through the complexities of dose translation, clinical trial planning, and regulatory compliance.
Let’s move toward a smarter, evidence-based approach to drug development together! 🚀